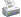Stoichiometric question
So, what factors keep folks tuning on the rich side? Is it because of unsafe temperatures? Insufficient fuel atomization? I know this is a straight-up newbie question to some of y'all, but after learning how the cats more or less burn off unburned fuel in the exhaust, I have to ask if it is possible to burn ALL of the gas in the combustion chamber, effectively negating the need to even use cats.
Am I onto sumpin, or has this been tried already?
So, what factors keep folks tuning on the rich side? Is it because of unsafe temperatures? Insufficient fuel atomization? I know this is a straight-up newbie question to some of y'all, but after learning how the cats more or less burn off unburned fuel in the exhaust, I have to ask if it is possible to burn ALL of the gas in the combustion chamber, effectively negating the need to even use cats.
Am I onto sumpin, or has this been tried already?
its to either make max power (slightly rich)
or to make max MPG (slightly lean)
the only reason it bounces around 14.67 stock, is that the OEMS want that perfect o2 burn half the time for the emissions. tuners generally dont worry about that as much.
Trending Topics
The Best V8 Stories One Small Block at Time
With this in mind I have been running 15.4:1 under 2400 RPM and 50kpa map for a few months with no issues for ECT temp, knock or otherwise so far.
Please explain how 16:1 will burn pistons during "max efficiency" mode

Simple answer is, you throw at it what you have in
surplus. For power, you can only get so much air in,
so maximize its use with excess fuel. For economy,
the opposite, give it air until you lose efficiency or
just your emissions certification.
i doubt a factory aluminum piston could handle the combustion temps associated with that ratio
So Jimmy, by your graph the stoichiometric ratio is pretty much where you get the most possible of both power and efficiency, right?
I need to read more, I can tell.
they do from the factory.... all over the world, except the USA... where the EPA has decided that its "cheating" to do that... along with some emissions BS that doesnt apply to real life, is based on theorys, and they made laws based on them anyway.
You need 14.7 parts O2 for each part of petrol in ideal world.
So CxHx + O2 => CO2 + H2O + Heat (Not sure what petrol is maybe C6H12)
Orange at low map there is no problem with 16:1 as mentioned Holdens have run it for years as lean cruise. Would you want to run it at WOT, no. But thats not what anyone is suggesting.
i didnt know we were talking about the world car industry
we dont have lean cruise here, so there are no safeguards in the PCM to run that lean AFR....
You need 14.7 parts O2 for each part of petrol in ideal world.
So CxHx + O2 => CO2 + H2O + Heat (Not sure what petrol is maybe C6H12)
Orange at low map there is no problem with 16:1 as mentioned Holdens have run it for years as lean cruise. Would you want to run it at WOT, no. But thats not what anyone is suggesting.
So it goes (assuming atmosphere is 21% oxygen and 79%Nitrogen.. all others are considered negligible for calcuation purposes)
C8H15 + (x)O2 + (x)3.76N2 --> (x)CO2 + (x) H20 + (x)NOx + (x)CO + Engergy!! + etc..
I dont feel like balancing equaitons so that is just a basic equation..
These are ECT, RPM, g/cyl and VSS.
Basically RPM and g/cyl is a map with AFR in it. Stock is lean 1200 RPM to 3200 RPM and 0.12g/cyl to 0.40g/cyl.
If you run open loop then you can make your own lean cruise by leaning up the bottom half of the commanded fuel table. I just did it under decel and at city speeds. After that its pretty much stoich till 80kpa. As mentioned ECT is fine.
BTW, re Stoich and emissions, the converter needs excess oxygen to burn/oxidize the engine's output of carbon monoxide and assorted hydrocarbons, but not to reduce its oxides of nitrogen, so calibrations do not run at a steady stoichiometric ratio, but rather 'dither' back and forth a tenth or two either side at ~10 cycles per second. This way, the converter can store oxygen during the lean excursions to use during the rich swings, and thus catalyze both oxidation and reduction reactions.
BTW2, 'gasoline' hasn't been a specific molecule for almost a century. Pump fuel formulas are myriad and ever-evolving and often consist of hundreds of compounds, which affects the actual stoichiometric ratio, although except in the case of significant alcohol content usually only by a tenth or two.