
Nitrous gauge Question?!!?
The pressure shouldn't get that high when filling the bottle unless the mother bottle is almost empty and they are just pumping away to get it filled. Then is might heat the bottle up to those pressures.
-Chris
Trending Topics
The Best V8 Stories One Small Block at Time
look at the diagram below.. at no point will increasing pressure turn any liquid to a gas ...
Gas PropertiesMolecular WeightMolecular weight : 44.013 g/mol
Solid phaseMelting point : -91 �C
Latent heat of fusion (1,013 bar, at triple point) : 148.53 kJ/kg
Liquid phaseLiquid density (1.013 bar at boiling point) : 1222.8 kg/m3
Liquid/gas equivalent (1.013 bar and 15 �C (59 �F)) : 662 vol/vol
Boiling point (1.013 bar) : -88.5 �C
Latent heat of vaporization (1.013 bar at boiling point) : 376.14 kJ/kg
Vapor pressure (at 20 �C or 68 �F) : 58.5 bar
Critical pointCritical temperature : 36.4 �C
Critical pressure : 72.45 bar
Gaseous phaseGas density (1.013 bar at boiling point) : 3.16 kg/m3
Gas density (1.013 bar and 15 �C (59 �F)) : 1.872 kg/m3
Compressibility Factor (Z) (1.013 bar and 15 �C (59 �F)) : 0.9939
Specific gravity (air = 1) (1.013 bar and 21 �C (70 �F)) : 1.53
Specific volume (1.013 bar and 21 �C (70 �F)) : 0.543 m3/kg
Heat capacity at constant pressure (Cp) (1.013 bar and 15 �C (59 �F)) : 0.038 kJ/(mol.K)
Heat capacity at constant volume (Cv) (1.013 bar and 15 �C (59 �F)) : 0.029 kJ/(mol.K)
Ratio of specific heats (Gamma:Cp/Cv) (1.013 bar and 15 �C (59 �F)) : 1.302256
Viscosity (1.013 bar and 0 �C (32 �F)) : 0.000136 Poise
Thermal conductivity (1.013 bar and 0 �C (32 �F)) : 14.57 mW/(m.K)
MiscellaneousSolubility in water (1.013 bar and 5 �C (41 �F)) : 1.14 vol/vol
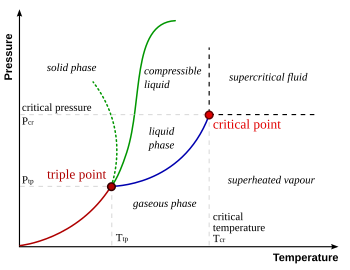
the dotted green line is the funky solid to fluid line of water.. without wich you wouldnt be able to skate (melting the ice by applying pressure and skating on a film of water)
Last edited by deuce_454; Jan 15, 2009 at 03:13 PM.













